PCOS is linked to changes in the level of certain hormones:
women with PCOS have either impaired glucose tolerance or Type 2 Diabetes . In addition, with Polycystic Ovarian Syndrome, high levels of insulin stimulate the ovaries to produce large amounts of testosterone (a male hormone), which can contribute to infertility by possibly preventing the ovaries from releasing an ovum each month.
Researchers have also found a link between Polycystic Ovarian Syndrome and other metabolic conditions such as high levels of obesity, LDL (the "bad" cholesterol) and high blood pressure, risk factors for coronary heart disease, as well as symptoms of Metabolic Syndrome. Also known as Syndrome X, this disorder can substantially increase the chances of developing Cardiovascular Disease. These findings substantially raise the bar on the seriousness of the condition and make it even more important that physicians correctly diagnose PCOS and recommend appropriate therapy.
Because insulin production of the pancreas is not balanced by Insulin Resistance, the conversion of food to energy is impaired increasing the amount of stored fats. When glucose cannot enter the cells efficiently, it remains in the blood stream, causing elevated blood sugar which is sent to the liver, where it converts to fat and stored throughout the body. which causes weight increase and ultimately can lead to obesity .
When the body takes in calories, it has either burn those calories for energy or convert them to fat. In women with Polycystic Ovarian Syndrome, Insulin Resistance encourages the storage of fat and the production of excessive amounts of the male hormone testosterone.
- Estrogen and progesterone, the female hormones that help a woman's ovaries release eggs.
- Androgen, a male hormone found in small amounts in women.
- insulin , a glucose (sugar) maintaining hormone.
- lifestyle choices .
women with PCOS have either impaired glucose tolerance or Type 2 Diabetes . In addition, with Polycystic Ovarian Syndrome, high levels of insulin stimulate the ovaries to produce large amounts of testosterone (a male hormone), which can contribute to infertility by possibly preventing the ovaries from releasing an ovum each month.
Researchers have also found a link between Polycystic Ovarian Syndrome and other metabolic conditions such as high levels of obesity, LDL (the "bad" cholesterol) and high blood pressure, risk factors for coronary heart disease, as well as symptoms of Metabolic Syndrome. Also known as Syndrome X, this disorder can substantially increase the chances of developing Cardiovascular Disease. These findings substantially raise the bar on the seriousness of the condition and make it even more important that physicians correctly diagnose PCOS and recommend appropriate therapy.
Because insulin production of the pancreas is not balanced by Insulin Resistance, the conversion of food to energy is impaired increasing the amount of stored fats. When glucose cannot enter the cells efficiently, it remains in the blood stream, causing elevated blood sugar which is sent to the liver, where it converts to fat and stored throughout the body. which causes weight increase and ultimately can lead to obesity .
When the body takes in calories, it has either burn those calories for energy or convert them to fat. In women with Polycystic Ovarian Syndrome, Insulin Resistance encourages the storage of fat and the production of excessive amounts of the male hormone testosterone.
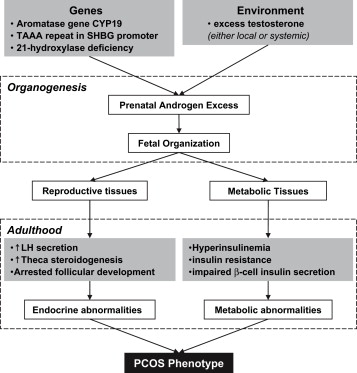
PCOS is a complex, heterogeneous disorder of uncertain aetiology. Both genes and the environment contribute to PCOS. Obesity, exacerbated by poor dietary choices and physical inactivity, worsens PCOS in susceptible individuals.There is strong evidence that there is a genetic component in many cases. Such evidence includes the familial clustering of cases, greater concordance in monozygotic compared with dizygotic twins and heritability of endocrine and metabolic features of PCOS.
The genetic component appears to be inherited in an autosomal dominant fashion with high genetic penetrance but variable expressivity in females; this means that each child has a 50% chance of inheriting the predisposing genetic variant(s) from a parent, and if a daughter receives the variant(s), then the daughter will have the disease to some extent.The genetic variant(s) can be inherited from either the father or the mother, and can be passed along to both sons (who may be asymptomatic carriers or may have symptoms such as early baldness and/or excessive hair) and daughters, who will show signs of PCOS. The allele appears to manifest itself at least partially via heightened androgen levels secreted by ovarian follicle theca cells from women with the allele. The exact gene affected has not yet been identified. The clinical severity of PCOS symptoms appears to be largely determined by factors such as obesity.
The genetic component appears to be inherited in an autosomal dominant fashion with high genetic penetrance but variable expressivity in females; this means that each child has a 50% chance of inheriting the predisposing genetic variant(s) from a parent, and if a daughter receives the variant(s), then the daughter will have the disease to some extent.The genetic variant(s) can be inherited from either the father or the mother, and can be passed along to both sons (who may be asymptomatic carriers or may have symptoms such as early baldness and/or excessive hair) and daughters, who will show signs of PCOS. The allele appears to manifest itself at least partially via heightened androgen levels secreted by ovarian follicle theca cells from women with the allele. The exact gene affected has not yet been identified. The clinical severity of PCOS symptoms appears to be largely determined by factors such as obesity.
The syndrome acquired its most widely used name due to the common sign on ultrasound examination of multiple (poly) ovarian cysts. These "cysts" are actually immature follicles, not cysts ("polyfollicular ovary syndrome" would have been a more accurate name). The follicles have developed from primordial follicles, but the development has stopped ("arrested") at an early antral stage due to the disturbed ovarian function. The follicles may be oriented along the ovarian periphery, appearing as a 'string of pearls' on ultrasound examination. There is also an increase in volume of ovary, especially due to increase in stroma.
Women with PCOS have higher GnRH, which in turn results in an increase in LH/FSH ratio.
A majority of patients with PCOS have insulin resistance and/or are obese. Their elevated insulin levels contribute to or cause the abnormalities seen in the hypothalamic-pituitary-ovarian axis that lead to PCOS. Hyperinsulinemia increases GnRH pulse frequency, LH over FSH dominance, increased ovarian androgen production,decreased follicular maturation, and decreased SHBG binding; all these steps contribute to the development of PCOS. Insulin resistance is a common finding among patients of normal weight as well as overweight patients.
In many cases PCOS is characterised by a complex positive feedback loop of insulin resistance and hyperandrogenism. In most cases it can not be determined which if anyof those two are responsible for causing pcos Experimental treatment with either antiandrogens or insulin sensitizing agents improves both hyperandrogenism and insulin resistance.
Adipose tissue possesses aromatase, an enzyme that converts androstenedione to estrone and testosterone to estradiol. The excess of adipose tissue in obese patients creates the paradox of having both excess androgens (which are responsible for hirsutism and virilization) and estrogens (which inhibits FSH via negative feedback) PCOS may be associated with chronic inflammation,with several investigators correlating inflammatory mediators with anovulation and other PCOS symptoms.
It has previously been suggested that the excessive androgen production in PCOS could be caused by a decreased serum level of IGFBP-1, in turn increasing the level of free IGF-I which stimulates ovarian androgen production, but recent data concludes this mechanism to be unlikely.
PCOS has also been associated with a specific FMR1 sub-genotype. The research suggests that women who have heterozygous-normal/low FMR1 have polycystic-like symptoms of excessive follicle-activity and hyperactive ovarian function.
Normal: less than or equal to 12 follicles. Borderline: 13-15 follicles. Mild: 16-30 follicles. Moderate: 30-50 follicles. Severe: more than 50 follicles, as a general guide - these women usually have Polycystic Ovarian Syndrome (PCOS), associated with infertility, increased facial hair, and a disturbance of hormone levels.
It takes a bit over a year, or 13 menstrual cycles, for a follicle to develop from dormancy up to the stage of releasing its eggs into the oviduct (the tube which carries the egg to the womb). The process requires a large group of undeveloped follicles to grow and be progressively weaned into one dominant preovulatory follicle. The dominant follicle of the cycle is said by some to be selected from a cohort of class five follicles (when they are 2 - 5mm), others say class 7 (greater than or equal to 10mm); it requires about 20 days to develop to the ovulatory stage from class 5. The eggreaches its maximum growth when the follicle is 0.2mm but ovulation doesn't occur until the follicle is about 16 - 20mm.
Typically around 20 follicles mature each month and only a single follicle is ovulated. If a maturing follicle does not become the dominant follicle, it is reabsorbed into the ovary (ovarian follicle atresia) and takes on a different cell role. In PCOS, this reabsorption does not occur. Lower levels of a chemical that triggers atresia (caspase 3) was found in PCOS patients. Higher levels of a chemical that defends against atresia was also found (cIAP-2)
Women with PCOS have higher GnRH, which in turn results in an increase in LH/FSH ratio.
A majority of patients with PCOS have insulin resistance and/or are obese. Their elevated insulin levels contribute to or cause the abnormalities seen in the hypothalamic-pituitary-ovarian axis that lead to PCOS. Hyperinsulinemia increases GnRH pulse frequency, LH over FSH dominance, increased ovarian androgen production,decreased follicular maturation, and decreased SHBG binding; all these steps contribute to the development of PCOS. Insulin resistance is a common finding among patients of normal weight as well as overweight patients.
In many cases PCOS is characterised by a complex positive feedback loop of insulin resistance and hyperandrogenism. In most cases it can not be determined which if anyof those two are responsible for causing pcos Experimental treatment with either antiandrogens or insulin sensitizing agents improves both hyperandrogenism and insulin resistance.
Adipose tissue possesses aromatase, an enzyme that converts androstenedione to estrone and testosterone to estradiol. The excess of adipose tissue in obese patients creates the paradox of having both excess androgens (which are responsible for hirsutism and virilization) and estrogens (which inhibits FSH via negative feedback) PCOS may be associated with chronic inflammation,with several investigators correlating inflammatory mediators with anovulation and other PCOS symptoms.
It has previously been suggested that the excessive androgen production in PCOS could be caused by a decreased serum level of IGFBP-1, in turn increasing the level of free IGF-I which stimulates ovarian androgen production, but recent data concludes this mechanism to be unlikely.
PCOS has also been associated with a specific FMR1 sub-genotype. The research suggests that women who have heterozygous-normal/low FMR1 have polycystic-like symptoms of excessive follicle-activity and hyperactive ovarian function.
Normal: less than or equal to 12 follicles. Borderline: 13-15 follicles. Mild: 16-30 follicles. Moderate: 30-50 follicles. Severe: more than 50 follicles, as a general guide - these women usually have Polycystic Ovarian Syndrome (PCOS), associated with infertility, increased facial hair, and a disturbance of hormone levels.
It takes a bit over a year, or 13 menstrual cycles, for a follicle to develop from dormancy up to the stage of releasing its eggs into the oviduct (the tube which carries the egg to the womb). The process requires a large group of undeveloped follicles to grow and be progressively weaned into one dominant preovulatory follicle. The dominant follicle of the cycle is said by some to be selected from a cohort of class five follicles (when they are 2 - 5mm), others say class 7 (greater than or equal to 10mm); it requires about 20 days to develop to the ovulatory stage from class 5. The eggreaches its maximum growth when the follicle is 0.2mm but ovulation doesn't occur until the follicle is about 16 - 20mm.
Typically around 20 follicles mature each month and only a single follicle is ovulated. If a maturing follicle does not become the dominant follicle, it is reabsorbed into the ovary (ovarian follicle atresia) and takes on a different cell role. In PCOS, this reabsorption does not occur. Lower levels of a chemical that triggers atresia (caspase 3) was found in PCOS patients. Higher levels of a chemical that defends against atresia was also found (cIAP-2)
References :
| genetics_of_polycystic_ovary_syndrome.pdf | |
| File Size: | 54 kb |
| File Type: | |
